Case of the Month ...
Case History
The patient is an 83-year-old Caucasian female who was found to have an antral mass and subsequently underwent partial gastrectomy in 2002. The diagnosis of Gastrointestinal Stromal Tumor (GIST) was rendered, and was based on the high mitotic count (>20/50 hpf), pleomorphism and necrosis. Surgical resection margins were negative and she was followed up by regular CT scans for five years. In September of 2007 an abdominal CT scan showed a 6.3 x 5.7 cm fibrotic mass in the right lobe of the liver. CT guided fine needle aspiration was performed to rule out metastatic GIST.
Diagnosis & Discussion
click on image for larger version
Figure 1
Image Figures:
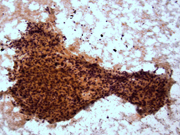
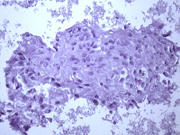
- Figure 1: FNA cytology smear, Papanicolaou stain, X 100
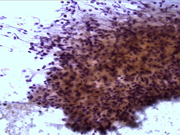
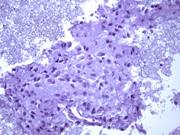
- Figure 2: FNA cytology smear, Papanicolaou stain, X 200
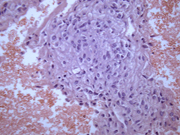
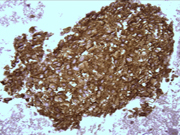
- Figure 3: FNA cytology smear, Papanicolaou stain, X 400
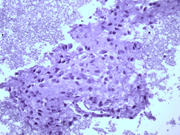
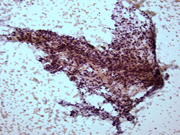
- Figure 4: FNA cell block, H & E, X 200
- Figure 5: FNA cell block, C-kit immunoperoxidase stain, X 200
- Figure 6: FNA cell block, Cytokeratin AE1/AE3 immunoperoxidase stain, X 200
- Figure 7: FNA cell block, Hep-Par1 immunoperoxidase stain, X 200
- Figure 8: FNA cell block, FNA cell block, PDGFR immunoperoxidase stain, X 200
Diagnosis: Metastatic epithelioid Gastrointestinal Stromal Tumor (GIST) to the liver (C-Kit negative and PDGFR positive).
Cytologic Findings:
- Hypercellular aspirate with cohesive fragments of polygonal cells.
- Individual cells were round to polygonal with moderate nuclear to cytoplasmic ratio.
- The cytoplasm was dense and rare small vacuoles were noted.
- Fine nuclear chromatin was identified.
- Small and inconspicuous nucleoli were identified.
- Endothelial cell lining small fragments of tumor cells was present.
- Necrotic debris in the background was identified.
Discussion: Gastrointestinal stromal tumors constitute the largest group of mesenchymal neoplasms in the gastrointestinal tract and due to their morphologic variability and unpredictable clinical behavior numerous studies have been conducted to establish diagnostic and prognostic criteria in these neoplasms. The cytologic features of both the epithelioid and spindle cell subtypes of GISTs are distinct and can be appreciated both on direct smears as well as cell block preparations. The cellularity can range from scant to moderate with elongated nuclear contours in the spindle cell variant, and polygonal in the epithelioid subtype, with fine chromatin and moderate to high nuclear to cytoplasmic ratio. When the epithelioid variant of GIST is encountered on a fine needle aspiration from the liver, the differential diagnosis of epithelioid leiomyosarcoma and angiosarcoma as well as metastatic adenocarcinoma enter the differential diagnosis. A panel of immunohistochemical stains is recommended therefore to confirm the diagnosis. Currently c-kit is considered the most specific marker in establishing the diagnosis of GIST. This can be challenging in cases where c-kit is negative. This diagnostic challenge was solved when studies confirmed that platelet-derived growth factor receptor (PDGFR) was overexpressed in c-kit negative tumors, and showed that there is in fact mutual exclusion of expression of these two proteins in GIST. Apart from being a diagnostic tool, the over expression of c-kit can predict response to tyrosinase inhibitors. It is well known that a small subset (4.7-6.2%) of GIST are c-kit negative and lack the characteristic point mutation. In these tumors, however mutation and over expression of PDGFR was noted both by immunohistochemical and molecular analysis. Interestingly, this small group of patient seems to respond to tyrosine kinase inhibitor treatment as well. These studies prompted the development and application of antibodies against PDGFR to detect c-kit negative GISTs by immunohistochemistry and to differentiate these neoplasms from other, morphologically similar gastrointestinal tumors.
In summary, fine needle aspiration specimens from the gastrointestinal tract, showing morphologic and cytologic features suggestive of a mesenchymal neoplasm should be worked up by a panel of immunohistochemical stains for exact classification and prognostic purposes. These ancillary studies can be performed on direct smears or cell block preparations. Criteria to establish the diagnosis of malignant GIST are applied on resection specimens and can be a diagnostic challenge on an FNA. Findings such as marked pleomorphism, epitheliod features, necrosis and numerous or atypical mitotic figures can indicate more aggressive behavior. Immunohistochemical stain for c-kit should be routinely performed and anti-PDGFR can be added in case of negativity. This approach can help triaging patients who would benefit from tyrosinase inhibitor (Gleevec) therapy.
References:
- Geisinger K. R., Stanley M.W., Raab S.S., Silverman J. F., Abati A. Modern Cytopathology, Churchill Livingstone 2004. pp: 478, 536-538.
- Padilla C., Saez A, Catala I. , Vidal A., Garcia L, Tolosa F., Andreu F.J., Combala N.: Fine-needle aspiration cytology diagnosis of metastatic gastrointestinal stromal tumor in the liver: A report of three cases. Diagnostic Cytopathology, 2002, Vol 27, No 5, pp: 298-302.
- Rossi G, Valli R, Bertolini F, Marchioni A, Cavazza A, Mucciarini C, Migaldi M, Federico M, Trentini G P, Sgambato A: PDGFR expression in differential diagnosis between KIT-negative gastrointestinal stromal tumours and other primary soft-tissue tumours of the gastrointestinal tract Histopathology 2005, 46, pp: 522-531.
- Gomes A L, Bardales R H, Milanezi F, Reis R M, Shcmitt F.: Molecular analysis of c-kit and PDGFRA in GISTs diagnosed by EUS. American Journal of Clinical Pathology 2007; 127: 89-96.
- Lozano M D, Rodriguez J, Algarra S M, Panizo A, Sola J, Pardo J: Fine-needle aspiration cytology and immunocytochemistry in the diagnosis of 24 gastrointestinal stromal tumors: A quick, reliable diagnostic method. Diagnostic Cytopathology, 2003; Vol 28, No3, pp: 131-135.
Acknowledgement: Submitted by Krisztina Hanley, M.D. and Momin T. Siddiqui M.D., FIAC. Emory University School of Medicine, Atlanta , GA.