Case of the Month ...
Case History
The patient is a 54 year old man who presented for the evaluation of a 3.1 cm. left parotid mass. The mass had been slowly growing for a period of approximately eleven months. On examination the mass was firm, poorly circumscribed and tender on palpation. A fine needle aspiration was performed.
Diagnosis & Discussion
click on image for larger version
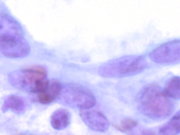
Figure 1
Image Figures:
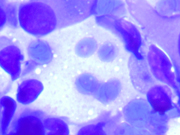
- Figure 1: Cytologic smear, Diff-Quik stain, X200
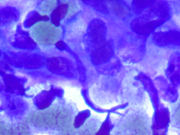
- Figure 2: Cytologic smear, Diff-Quik stain, X400
- Figure 3: Cytologic smear, Diff-Quik stain, X 400
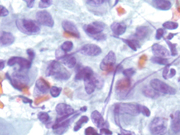
- Figure 4: Cytologic smear, Papanicolaou stain, X 200
- Figure 5: Cytologic smear, Papanicolaou stain, X 200
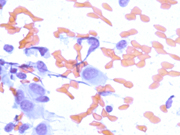
- Figure 6: Cytologic smear, Papanicolaou stain, X 600
Diagnosis: Myoepithelial carcinoma of the salivary gland.
Cytologic Findings:
- Hypercellularity with single cells and large tissue fragments.
- The tissue fragments appear three-dimensional with considerable nuclear overlapping and crowding.
- Metachromatic stromal fragments are occasionally seen intermixed with the neoplastic cells.
- Neoplastic cells consist predominantly of spindle cells with variable amounts of wispy pale cytoplasm with frayed cytoplasmic borders. Scattered epithelioid and plasmacytoid neoplastic cells can also be present.
- The nuclei range from oval to elongated to spindle shaped and vary considerably in size.
- The chromatin is coarsely granular and nucleoli are evident.
- Binucleated and multinucleated tumor cells and rare mitotic figures can also be present.
Discussion: Myoepithelial carcinomas (or malignant myoepitheliomas) are rare malignant salivary gland neoplasms in which the tumor cells show myoepithelial differentiation. The entity was first described by Stromeyer et al. in 1975. The tumor was included in the WHO classification of salivary gland neoplasms as a distinct clinicopathologic entity in 1991. About 60% to 70% of myoepithelial carcinomas develop in a benign mixed tumor (carcinoma ex pleomorphic adenoma), and the remainder arise de novo. Histologically, malignant myoepitheliomas are composed of one or several cell types: spindle, plasmacytoid, epithelioid, and clear cells. Frequently, one of the cell types predominates. The neoplastic cells grow either as multiple nodules or as large solid sheets separated by variable amounts of intervening hyaline or myxoid stroma. By far, the multinodular growth pattern is more prevalent. The cytologic features generally reflect the histology. The cytologic smear can show spindle, epithelioid or plasmacytoid cells. Scant fragments of metachromatic stroma intermixed with the neoplastic cells might be observed in the cytologic specimens of malignant myoepithelioma, regardless of the composition of the cell types.
Tumor infiltration into adjacent normal tissue is the most important histologic feature that distinguishes malignant from benign myoepithelial neoplasms and should be considered the minimum requirement for myoepithelial carcinoma. However, this feature is difficult if not impossible to appreciate with cytologic specimens and therefore cannot be used as a requirement for malignancy in FNA. Other features that signify malignancy, such as cytologic atypia, pleomorphism, necrosis, and mitotic activity, might not all be present in each case. Hence a diagnosis of malignancy may not be always possible based on cytologic morphology alone, particularly in cases where the diagnosis of malignancy is based solely on an infiltrative growth pattern rather than on cytologic atypia and/or mitotic activity. Proliferative activity by immunohistochemical staining with antibody to MiB1 (Ki-67) might be helpful in the differential diagnosis between benign and malignant myoepithelial neoplasms. A Ki-67 labeling index of more than 10% is reportedly diagnostic of myoepithelial carcinoma.
In view of its diverse cytologic presentations, myoepithelial carcinoma raises a variety of neoplasms in the differential diagnosis. Benign mixed tumors should be included in the differential diagnosis of myoepithelial carcinoma, particularly when the stromal
component of the myoepithelial carcinoma is conspicuous, resulting in biphasic pattern, and when there is a lack of significant cytologic atypia of the neoplastic cells. This challenge is further complicated by the fact that 60% to 70% of myoepithelial
carcinomas arise in benign mixed tumors. The findings of abundant chondromyxoid matrix and the lack of significant cytologic atypia and/or mitotic figures favor a benign mixed tumor over a Myoepithelial carcinoma. Oncocytic adenoma might also be confused with a myoepithelial neoplasm displaying a plasmacytoid cell appearance. The distinction between the two entities lies in that oncocytes have a granular cytoplasm and centrally located nuclei, whereas plasmacytoid myoepithelial cells have nongranular
cytoplasm and eccentrically located nuclei. Neoplastic spindle cells might be seen in Myoepithelial tumors but not oncocytic neoplasms. As a great mimicker, amelanotic melanoma should be included in the differential diagnosis. A loosely cohesive pattern of spindle or polygonal tumor cells, plasmacytoid appearance, prominent nucleoli, and binucleation are features of myoepithelial neoplasms that are also seen in melanoma. Conversely, intranuclear pseudoinclusion, a feature associated with melanoma, has also been reported in Myoepithelial carcinoma. The differential diagnosis also includes
both benign and malignant mesenchymal neoplasms, such as tumors of smooth muscle, fibroblasts, or peripheral nerve sheath, particularly when neoplastic spindle cells predominate.
he use of ancillary studies, such as immunohistochemistry, may be helpful in the recognition of the myoepithelial differentiation of the neoplastic cells. Neoplastic cells are immunoreactive to cytokeratin, S-100 protein, and smooth muscle actin, supporting a myoepithelial origin. Other myoepithelial markers include GFAP and calponin. The latter is a relatively new smooth muscle antibody that has been shown to be a specific and fairly sensitive marker of myoepithelial cells in salivary gland neoplasms.
References:
Seifert G. Histological typing of salivary gland tumors. In: World Health Organization International Histological Classification of Tumours. Berlin : Springer-Verlag, 1991.
Chow LT, Chow WH, Lee JC. Monomorphic epithelioid variant of malignant myoepithelioma of the parotid gland: cytologic features in fine needle aspiration (FNA). Cytopathology 1996;7:279-87.
DiPalma S, Alasio L, Pilotti S. Fine needle aspiration (FNA) appearances of malignant myoepithelioma of the parotid gland. Cytopathology 1996;7:357-65.
Torlakovic E, Ames ED, Manivel JC, Stanley MW. Benign and malignant neoplasms of myoepithelial cells: cytologic findings. Diagn Cytopathol 1993;9:655-60.
Stromeyer FW, Haggitt RC, Nelson JF, Hardman JM. Myoepithelioma of minor salivary gland origin. Light and electron microscopical study. Arch Pathol Lab Med 1975;99: 242-5.
Chhieng DC, Paulino AF. Cytology of Myoepithelial Carcinoma of the Salivary Gland: A Study of Four Cases. Cancer (Cancer Cytopathol) 2002;96:326.
Acknowledgment: Submitted by Momin T. Siddiqui M.D., FIAC, Emory University Hospital , Atlanta , GA.