Case of the Month ...
Case History
A 41 year old female with no significant medical history had her annual screening mammography. The mammogram demonstrated two new irregular stellate masses in the left breast at 10 o'clock and at 1 o'clock with calcifications. The lesions were assigned a BI-RADs 5 category as highly suspicious for malignancy. An ultrasound exam showed irregular masses, both taller than wide, with architectural distortion of the adjacent tissue, measuring 1.1 and 1 cm respectively. An ultrasound guided aspiration biopsy of both lesions followed by a core biopsy was performed.
Diagnosis & Discussion
click on image for larger versionImage Figs:
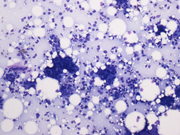
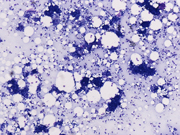
- Figure 1. FNA smears, Diff-quik stain, X100
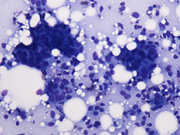
- Figure 2. FNA smears, Diff-quik stain, X200
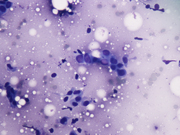
- Figure 3. FNA smears, Diff-quik stain, X400
- Figure 4. FNA smears, Diff-quik stain, X400
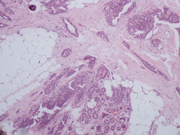
- Figure 5. Core biopsy, Hematoxylin and eosin, X100
Questions:
- What is the most accurate cytologic diagnosis?
- Adenocarcinoma
- Fibroadenoma
- Fibrocystic changes with atypia
- Cyst contents
- Three-dimensional cellular clusters
- Flat sheets of epithelial cells
- Presence of bare oval nuclei
- Loss of cellular cohesion
- An infiltrating pattern is a characteristic feature of the following lesion:
- Radial scar
- Sclerosing adenosis
- Lactating adenoma
- Fibrocystic changes
- Excision of the radial scar is recommended due to:
- Distortion of surrounding tissue
- Carcinoma and atypia is found on excision in a significant percentage of cases diagnosed as radial scar on core biopsy
- Associated florid usual ductal hyperplasia
- Associated mucocele like lesions
Discussion:
Aspiration smears were highly cellular with cohesive clusters and sheets of ductal epithelial cells along with groups of mildly enlarged ductal epithelial cells with increased nuclear to cytoplasmic ratios, mild variation of nuclear size, and inconspicuous nucleoli. Cells had a small to moderate amount of amphophilic cytoplasm. No significant loss of cohesion within the groups of ductal cells was noticed. Scattered bare oval myoepithelial cell nuclei, histiocytes, thin proteinaceous material, and stromal fragments were present. The on-site assessment diagnosis was fibrocystic changes, proliferative type, with atypia. A core biopsy was recommended and was immediately performed. It revealed a proliferation of tubular structures with round and slit-like lumens lined by ductal epithelial cells without significant atypia. The tubules were arranged around fibrillary elastoid material in an infiltrative pattern. Some of the lumens demonstrated florid usual ductal hyperplasia. Cystically dilated ducts lined by apocrine metaplastic cells were also present. Microcalcifications were noted within the ductal lumens. Myoepithelial cells were distinguishable around the tubular structures on H&E sections and highlighted with calponin and p63 immunostains. A diagnosis of radial sclerosing lesion with associated florid duct hyperplasia, sclerosing adenosis, and apocrine metaplasia was made.
Cytomorphologic features of radial scar are relatively non-specific and often overlap with adenosis, sclerosing adenosis, fibroadenoma, and fibrocystic change, although their mammographic and ultrasonographic appearance differs. The aspirates vary from scant to very cellular with cohesive clusters of ductal epithelial cells arranged in flat sheets. Tubular structures, acinar and papillary arrangements of ductal epithelial cells have also been described. Bare oval myoepithelial cell nuclei are usually seen. Histiocytes, apocrine cells, and fibrillary metachromatic stromal material are usually present in the background. Nuclear atypia and loss of cell cohesion may be seen in the smears leading to a core or an excisional biopsy to exclude malignancy. Histologically, radial sclerosing lesions are characterized by a central area of fibroelastotic tissue with radiating ducts spreading into the surrounding tissue in an infiltrative pattern. The ducts often have an elongated appearance and compressed slit-like lumens. Dilated ducts with various amounts of epithelial hyperplasia along with adenosis may be present.
Differential diagnosis of a radial scar includes fibrocystic changes, adenosis and well differentiated ductal carcinoma. Radiographically and pathologically it is difficult to distinguish a radial scar from carcinoma due to its stellate configuration. A specific diagnosis of a radial scar by FNA in most cases is not possible due to overlapping features. Some data suggests that radial scar may be an independent risk factor for the development of breast carcinoma. Well differentiated carcinoma, including tubular carcinoma often shows angular tubular structures with only mild nuclear atypia and without the loss cell cohesion on cytology. Tissue sections demonstrate infiltrating ductal structures without myoepithelial cells. The ducts are surrounded by fibrotic desmoplastic stromal tissue that may simulate the fibroelastotic stroma of a radial scar. The ducts have angulated appearance and the lumens are characteristically open. FNA smears of fibrocystic changes and adenosis usually show cohesive clusters of ductal epithelial cells with a various amount of sclerotic stroma. Bare oval nuclei of myoepithelial cells may be present. A well circumscribed radiographic appearance is characteristic. Histologic sections of fibrocystic changes show cystically dilated ducts with frequent apocrine metaplasia and dense sclerotic stroma. Sclerosing adenosis shows a disordered proliferation of acinar and ductal epithelial cells, myoepithelial cells and intralobular stroma that result in expansion and distortion of lobules. This lobulocentric pattern is characteristic for adenosis and serves as a main diagnostic feature. The presence of a myoepithelial cell layer on either H&E stain or by immunohistochemistry (p63, calponin or SMA) helps to exclude a tubular carcinoma and highlights the myoepithelial cell layer in radial scar and sclerosing adenosis. Surgical excision is recommended when the imaging findings are compatible with a radial scar. Core needle biopsy for these lesions is problematic due to the potential for sampling error and the possible confusion with invasive carcinoma on histologic assessment of limited samples According to some studies up to 60% of the cases may show atypical ductal hyperplasia at excision and up to 40% of the cases may show carcinoma. Carcinoma is usually found at the periphery of the radial scar at excision, an important issue to consider when sampling by core needle biopsy.
Answers
- c
- d
- a
- b
REFERENCES
- Levine P, Cangiarella J. Cytomorphology of benign breast disease. Clinics in laboratory medicine. 2005; 25(4):689-712.
- Jacobs T, Byrne C, Colditz G et al. Radial scars in benign breast biopsy specimens and the risk of cancer. N Engl J Med 1999; 340:430-436.
- Lopez-Medina A, Cintora E, Mugica B et al. Radial scars diagnosed at stereotactic core-needle biopsy: surgical biopsy findings. Eur Radiol 2006;16:1803-1810.
- Sloane J, Mayers MM. Carcinoma and atypical hyperplasia in radial scars and complex sclerosing lesions: importance of lesion size and patient age. Histopathology 1993;23:225-231.
- Cawson J, Malara F, Kavanagh A et al. Fourteen gauge needle CNB of mammographically evident radial scars. Is excision necessary? Cancer 2003;97:345-351.
Contributed by Alexander Finkelstein, DO and Joan Cangiarella, MD
NYU School of Medicine
Department of Pathology
New York , NY