Case of the Month ...
Clinical History:
A 39 year-old female patient was found to have a pancreatic mass (hypo and hyper -echoic) on ultrasound measuring 4.2 x 2.7 cm. The mass was located in the head of the pancreas. Endoscopic ultrasound-guided fine needle aspiration (EUS-FNA) of the pancreatic mass was performed.
Diagnosis & Discussion
click on image for larger version
Image Figs:
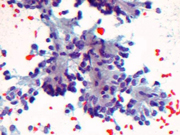
Figure 1: FNA cytology, Rapid Romanowsky stain, X200
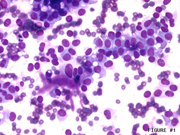
Figure 2: FNA cytology, Pap stain, X200
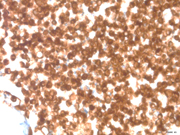
Figure 3: FNA cytology, Beta Catenin stain (immunostain), X200
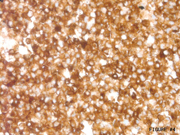
Figure 4: FNA cytology, CD 10 stain (immunostain), X200
Questions:
What is the best differential diagnosis?
Carcinoma and melanoma
Lymphoma and autoimmune pancreatitis
Solid-pseudopapillary neoplasm of pancreas and neuroendocrine tumor
Sarcoma and mesothelioma
Which are the typical cytomorphologic features of this tumor?
Pleomorphic atypical cells, single and in groups
Single plasmacytoid small cells with salt and pepper chromatin pattern and scant cytoplasm and small intra cytoplasmic vacuoles
Single plasmacytoid cells and groups of cells forming papillary groups and large intra cytoplasmic vacuoles
Single plasmacytoid cells with prominent macro nucleoli, bi-nucleate cells and intra nuclear cytoplasmic inclusions
What ancillary studies will help to arrive at the diagnosis?
Flow Cytometry
Next gene sequencing
Immunostains
Which are the typical immunophenotypic features of this tumor?
Mucicarmine +
Synaptophysin+, chromogranin+, E-Cadheirn+ PR- Beta-catenin -
S-100+, HMB 45-ve, MART-1 +
E-Cadherin-, PR+, Beta-catenin+, CD10 +, Synaptophysin +/-Chromogranin+/-
Discussion:
The origin of solid pseudopapillary neoplasm of the pancreas (SPN) remains largely unknown. This tumor is common in young females and is usually located in the head or tail of the pancreas. The sign and symptoms include abdominal pain, 1 nausea, vomiting, fever, weight loss, and jaundice. This tumor is often found incidentally via imaging studies. Radiologically, this tumor exhibits solid and cystic areas, often with areas of hemorrhage. SPN can have a radiologic appearance and clinical features that overlap with pancreatic pseudocyst and radiologic/cytologic appearances that overlap with other pancreatic neoplasms, including acinar cell carcinoma, mucinous neoplasms, and pancreatic endocrine neoplasm (PEN). Hence,It is important to establish a preoperative diagnosis of SPN because these tumors exhibit a different biologic behavior, and their management protocols may differ accordingly.
Endoscopic ultrasound guided fine-needle aspiration (EUS-FNA) is a very useful method for diagnosing pancreatic lesions. Cytomorphologic features of SPN includes a mixture of papillary groups and single plasmacytoid cells in the background (Images 1 and 2). Magenta colored matrix and fibrovascular cores are often identified in the papillary groups. The nuclei are elongated and they often shows grooves. Large cytoplasmic vacuoles can also be seen which helps in distinguishing this entity from pancreatic neuroendocrine neoplasm (PEN). FNA of PEN shows single plasmacytoid cells with salt and pepper chromatin pattern. Small cytoplasmic vacuoles can also be seen and helps in the differential diagnosis from SPN. However, their distinction in cytologic samples can be challenging owing to overlapping morphologic features. In such circumstances, ancillary stains can provide much needed supportive evidence. However, SPN has been noted to demonstrate neuroendocrine differentiation on immunohistochemical stains and on electron microscopic studies. Thus, relying only on evidence of neuroendocrine differentiation on cytologic samples can lead to diagnostic errors. To add to this dilemma, FNA samples may sometimes not have enough cellularity to perform a large battery of immunohistochemical stains. Therefore, antibodies need to be chosen judiciously to best use limited cytologic material to distinguish among these entities.
In general, SPN is E-Cadherin (no cytoplasmic membrane staining), Beta-Catenin+ (Image 3), CD10+ (Image 4), PR+, Chromogranin/Synaptophysin+/-. PEN is E-Cadherin + (cytoplasmic membrane staining+), CD10-, Chromogranin/Synaptophysin+/-. Thus, use of a limited immunohistochemical panel that includes Beta-Catenin, E-Cadherin and CD10 is very useful to differentiate SPN and PEN.Answer Key:
C
C
C
D
REFERENCES
Adams AL, Siegal GP, Jhala NC. Solid pseudopapillary tumor of the pancreas: a review of salient clinical and pathologic features. Adv Anat Pathol. 2008;15:3945
Klimstra DS, Wenig BM, Heffes CS. Solid-pseudopapillary tumor of the pancreas: a typically cystic carcinoma of low malignant potential. Semin Diagn Pathol. 2000;17:6680.
Dong DJ and Zhang SZ. Solid-pseudopapillary tumor of the pancreas: CT and MRI features of 3 cases. Hepatobiliary Pancreat Dis Int. 2006;5:300304.
Machado MC, Machado MA, Bacchella T, Bacchella T, Jukemura J, Almeida JL, Cunha JE. Solid pseudopapillary neoplasm of the pancreas: distinct patterns of onset, diagnosis, and prognosis for male versus female patients. Surgery. 2008;143:2934.
Bhanot P , Nealon WH, Walser EM, Bhutani MS, Tang WW, Logroño R. Clinical, imaging, and cytopathological features of solid pseudopapillary tumor of the pancreas: a clinicopathologic study of three cases and review of the literature. Diagn Cytopathol. 2005;33:421428.
Jhala NC , Jhala DN, Chhieng DC, Eloubeidi MA, Eltoum IA. Endoscopic ultrasoundguided fine-needle aspiration: a cytopathologist's perspective. Am J Clin Pathol. 2003;120:351367.
Pitman MB, Deshpande V. Endoscopic ultrasoundguided fine needle aspiration cytology of the pancreas: a morphological and multimodal approach to the diagnosis of solid and cystic mass lesions. Cytopathology. 2007;18:331347.
Jhala N , Siegal GP , Jhala D. Fine-needle aspiration: a powerful modality in the preoperative diagnosis of solid pseudopapillary neoplasm of the pancreas. Pathol Case Rev. 2007;12:170176.
Jhala N , Siegal GP , Jhala D. Large, clear cytoplasmic vacuolation: an under-recognized cytologic clue to distinguish solid pseudopapillary neoplasm of the pancreas from pancreatic endocrine neoplasm on fine-needle aspiration. Cancer. 2008;114:249254.
Bardales RH, Centeno B, Mallery JS, Lai R, Pochapin M, Guiter G, Stanley MW. Endoscopic ultrasoundguided fine-needle aspiration cytology diagnosis of solid-pseudopapillary tumor of the pancreas: a rare neoplasm of elusive origin but characteristic cytomorphologic features. Am J Clin Pathol. 2004;121:654662.
Matsunou H, Konishi F . Papillary-cystic neoplasm of the pancreas. A clinicopathologic study concerning the tumor aging and malignancy of nine cases. Cancer. 1990;65:283291.
Aydiner F, Erinanc H, Savas B, Erden E, Karayalcin K. Solid pseudopapillary tumor of the pancreas: emphasis on differential diagnosis from aggressive tumors of the pancreas. Turk J Gastroenterol. 2006;17:219222.
Burford H, Baloch Z, Liu X, Jhala D, Siegal GP , Jhala N . E-cadherin/beta-catenin and CD10: a limited immunohistochemical panel to distinguish pancreatic endocrine neoplasm from solid pseudopapillary neoplasm of the pancreas on endoscopic ultrasound-guided fine-needle aspirates of the pancreas. Am J Clin Pathol. 2009;132(6):831-9.
Contributed by:
Darshana Jhala, M.D.
Director of Anatomic Pathology
Philadelphia VA Medical Center
Associate Professor of Pathology
Department of Pathology
University of Pennsylvania