Case of the Month ...
Clinical History:
A 78 year old male presented for a fine needle aspiration of a thyroid nodule which was originally discovered four years prior. Thyroid function tests were reportedly within normal limits and no family history of thyroid disease was noted. The patient had no history of radiation to the head and neck area, no symptoms at the base of the neck and not had the nodule previously biopsied.\
The patient's sonographic report commented on a multinodular thyroid gland with 4 subcentimeter nodules and a dominant nodule, 19mm, in the right lobe, upper pole. This dominant nodule was palpable at the time of aspiration and two multidirectional passes using 25 gauge needles were performed.
Diagnosis & Discussion
click on image for larger version
Image Figs:
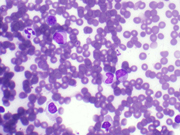
- FNA cytology smear of the right lobe thyroid mass, Diff-Quik stain, x40
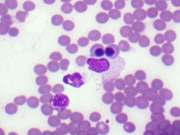
- FNA cytology smear of the right lobe thyroid mass, Diff-Quik stain, x40
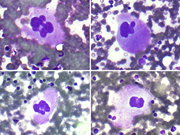
- FNA cytology smear of the right lobe thyroid mass, Diff-Quik stain, x40
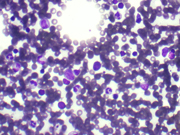
- FNA cytology smear of the right lobe thyroid mass, Diff-Quik stain, x40, composite
Questions:
What is the most likely cytologic diagnosis?
- Anaplastic carcinoma
- Medullary carcinoma
- Hashimoto's thyroiditis
- Myeloid sarcoma
- Trilinear hematopoiesis
- For this lesion, which of the following is a characteristic feature?
- Red granules
- Monoclonal cells
- Progressive maturation
- Plasmacytoid cells
- Which of the following is true for this lesion?
- It occurs with inadvertent intrathyroidal lymph node aspiration
- Immunohistochemical stains are not necessary to make the diagnosis
- It is seen most commonly in young patients
- Flow cytometric analysis is helpful in making the diagnosis
- Among the choices, the least common site to encounter this lesion is:
- Liver
- A lymph node
- Thyroid
- Spleen
Discussion:
Trilinear hematopoiesis (TH) can occur in thyroidal aspirations by one of three mechanisms: as mass-forming extramedullary hematopoiesis (EMH), as marrow elements within thyroidal metaplastic bone or as marrow elements in aspirated ossified neck cartilage.
Extramedullary hematopoiesis is a dynamic process of blood cell development occurring outside the confines of the medullary cavity of bone often in the settings of hypoxia, inflammation and bone marrow failure. Several case reports exist of EMH in various organs mimicking a mass and being discovered on fine needle aspiration. Although liver, spleen and lymph nodes are the most common sites, thyroid EMH can also occur. In a 2008 review, Westhoff et al. found that EMH of the thyroid most often occurs in older patients and is most likely secondary to a myelophthisic disease, such as myelofibrosis or metastatic bone marrow disease. Some reported cases of EMH in the thyroid have been attributable only to chronic anemia and still other cases have had no definable underlying cause. Since EMH has been described in nearly every organ system and can present as a mass-like lesion, recognition of benign trilinear hematopoiesis from aspirations of mass' lesions from any site is an important consideration.
Another possibility for thyroid trilinear hematopoiesis, although vanishingly rare, is a thyroid mass that is metaplastic bone formation within which there are foci of hematopoiesis. Regressive changes occur frequently within nodular goiters, often leading to dystrophic calcification which potentially, yet very rarely, progress to bone formation within the thyroid. Only a couple cases, mostly in young women, have ever been reported. To our knowledge, no such cases have been discovered through fine needle aspiration.
The last possibility for trilinear hematopoiesis in a thyroidal aspiration is the inadvertent sampling of ossified neck cartilage containing hematopoietic elements, as was first suggested in two cases described by Gay et al. in 1985. It is well known that portions of cricoid, arytenoid and thyroid cartilages ossify with increasing age. Within this ossification, foci of hematopoiesis can occur and thus be susceptible to aspiration. A lack of evidence for ossification within the thyroid, as on radiographic imaging, speaks against this possibility on fine needle aspiration.
The FNA smears of our case showed numerous lymphoid cells, many large and immature appearing in a background of blood. These cells represent the developing myeloid precursors. In addition, there were abundant cells with eccentric, dark, pyknotic nuclei and gray cytoplasm representative of normoblasts. Finally, the occasional large, multinucleated and multilobated cells with moderate cytoplasm that were seen are characteristic of megakaryocytes. A few, scattered mitotic figures were noted. No definitive follicular cells or colloid was seen.
At the time of aspiration, an on-site adequacy assessment was done and the material was ample for a descriptive diagnosis. Given the abundant lymphoid component, the possibility of a florid chronic lymphocytic (Hashimoto's) thyroiditis was initially considered. After a full review of the material, a diagnosis of trilinear hematopoiesis was made. A comment was provided that the aspiration may represent a mass-forming site of EMH or could be unintentionally aspirated ossified cartilage. Based on the provided ultrasound report, metaplastic bone formation was not entertained.
When trilinear hematopoiesis can be recognized by cytomorphologic features, the diagnosis is relatively straightforward. Cytomorphology of trilinear hematopoiesis mimics normal bone marrow aspirates and includes erythroid, myeloid and megakaryocytic cell lines. The hematopoietic precursors of each of the three lineages show progressive maturation as well as cytoplasmic and nuclear synchrony. If not recognized, trilinear hematopoiesis can be mistaken for malignancy. The cytologic differential diagnosis of TH most often includes lymphoma, myeloid sarcoma and anaplastic carcinoma. All of these entities portend far worse prognoses. The immaturity and size of the precursor myeloid cells can raise suspicion for lymphoma. Myeloid sarcoma will also have the atypical, immature granulocytic cells, similar to TH. However, megakaryocytes and erythroid precursors will not be apparent in either lymphoma or myeloid sarcoma. In contrast, anaplastic carcinoma often has bizarre, large atypical cells which can occur singly and mimic the megakaryocytes of TH. If any doubt, some authors suggest a keratin immunostain to help highlight anaplastic cells.
In conclusion, we describe a case of trilinear hematopoiesis in a thyroid nodule aspiration. Clinical and radiographic correlation can help favor true extramedullary hematopoiesis versus chance ossified cartilage aspiration.
Answer Key:
• E
• C
• B
• C
REFERENCES
- Kim, C. Homeostatic and pathogenic extramedullary hematopoiesis. J Blood Med. 2010; 1: 1319.
- Policarpio-Nicolas M, Bregman S, Ihsan M et al. Mass-forming extramedullary hematopoiesis diagnosed by fine-needle aspiration cytology. Diagn Cytopathol. 2006 Dec;34(12):807-11.
- Leung N, Yazdi H, Walsh S, et al. Extramedullary Hematopoiesis in a Thyroid Nodule. Endocrinologist. 8(5):369-372, September/October 1998.
- Westhoff CC, Karakas E, Dietz C et al. Intrathyroidal hematopoiesis: a rare histological finding in an otherwise healthy patient and review of the literature. Langenbecks Arch Surg. 2008 Sep;393(5):745-9.
- Pontikides N, Botsios D, Kariki E, et al. Extramedullary hemopoiesis in a thyroid nodule with extensive bone metaplasia and mature bone formation. Thyroid. 2003 Sep;13(9):877-80.
- Gay JD, Bjornsson J, Goellner JR. Hematopoietic cells in thyroid fine-needle aspirates for cytologic study: report of two cases. Mayo Clin Proc. 1985 Feb;60(2):123-4.
- Fassina A, Fedeli U, Borsato S. Extramedullary hematopoiesis of the thyroid gland diagnosed by FNA cytology. A case report. Acta Cytol. 1999 Nov-Dec;43(6):1181-3.
Contributed by:
Jay C. Zeck, M.D.
MedStar Georgetown University Hospital
Department of Pathology
Washington, D.C.Anne Busseniers, M.D.
Metropolitan Fine Needle Aspiration Services
Washington, D.C.