Case of the Month ...
Clinical History:
The patient was a 48 year-old female who presented with a right chick mass. The mass has been slowly growing for one year. On physical exam, a 3.5 cm firm right parotid mass was centered along the anterior border of the gland. The mass was fixed with erythematous changes overlying the cheek skin. MRI showed a 3.5 cm right parotid mass and demonstrated well-defined margins. No pathologically enlarged lymph nodes were seen. Given the well-define margins, the possibility of benign mixed tumor was high in the differential diagnoses, however more aggressive etiologies such as carcinoma ex pleomorphic adenoma, adenoid cystic carcinoma, mucoepidermoid carcinoma cannot be ruled out. A FNA of the parotid mass was performed.
Diagnosis & Discussion
click on image for larger version
Images 1-7:
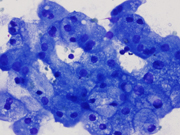
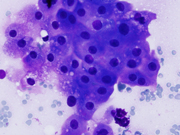
Figure 1. Diff Quik-stained smears. Medium magnification.
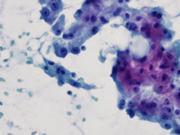
Figure 2. Diff Quik-stained smears. High magnification.
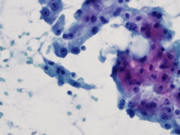
Figure 3. Pap-stained smears. Medium magnification.
Figure 4. Pap-stained smears. Medium magnification.
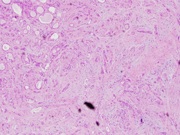
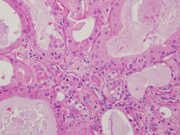
Figure 5. Resection, H&E-stain. Low magnification.
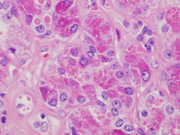
Figure 6. Resection, H&E-stain. High magnification.
Figure 7. Resection, H&E-stain. High magnification.Figure 1. Aggregates of epithelial cells showing hybrid of abundant granular cytoplasm and sebaceous-like vacuoles. Background of eosinophilic debris.
Figure 2. Aggregates of epithelial cells with sebaceous-like cytoplasm.
Figure 3. Aggregates of apocrine cells with round nucleus and prominent nucleoli.
Figure 4. Aggregates of epithelial cells showing hybrid features of sebaceous and apocrine cytoplasm.
Figure 5. Sclerosing fibrosis with entrapped ducts and dilated cysts.
Figure 6. Ductal epithelium with apocrine metaplasia and eosinophilic debris.
Figure 7. Acinar cells with granular cytoplasm and eosinophilic debris.Questions:
- The most likely diagnosis is:
- Sialadenitis
- Warthin’s tumor
- Oncocytoma
- Acinic cell carcinoma
- Sclerosing polycystic adenosis
- The nature of this lesion is
- More common in male patients
- A slow growing mass of unclear etiology
- Malignant neoplasm
- Only be seen in major salivary glands
- More common in minor salivary glands
- Cytomorphologic and histologic features are reminiscent of diseases from which organ:
- Prostate
- Breast
- Pancreas
- Skin
- What are the cytoplasmic qualities commonly found in this lesion
- Apocrine
- Vacuolated
- Apocrine and vacuolated
- Mucin
- Squamoid
Discussion:
Sclerosing polycystic adenosis (SPA) is a rare entity of the salivary gland that presents as a slow growing mass. Smith et al first described it in 1996 in a series consisting of 9 cases.(1) Afterwards, a total of 19 original case studies comprising over 50 cases have been published in the English literature. According to a review by Petersson, eighty percent of SPA affect parotid gland and is mostly unifocal though involvement of minor salivary glands, as well as multifocality may rarely be encountered. This condition affects male and female equally with a wider age spectrum of 9 to 84 years old; the size of the reported SPA ranges from 0.7 cm to 12 cm; recurrence may occurred in up to 19% of the cases. Although SPA often presented as an isolated finding, it may be associated with other entities including pleomorphic adenoma, oncocytoma, Warthin’s tumor. It is a histologically complex entity that consists of enlarged and distorted lobules, crowded acini with apocrine metaplasia, prominent stromal fibrosis and is reminiscent of fibrocystic disease of the breast. Chronic inflammation, myxoid stroma and various degrees of intraductal proliferations may present. Immunoprofiles reveal positivity for AE1/AE3 and Cam5.2 in both ductal and acinar cells while GCDFP-15 is strongly expressed in the acinar component; Consistent but variable expression of ER and PR is detected in ductal and acinar cells while Her-2/neu is negative.(2) The etiology of SPA is still unclear and the nature of this lesion was initially believed to be pseudoneoplastic inflammatory sclerosing process. However, monoclonality has been detected by human androgen receptor assay in SPA cases with epithelial atypia.(3) Further, a single case of invasive carcinoma arising from SPA has been recently reported.(4)
Cytology:
In the current case, Diff Quik-stained smears were hypercellular with abundant granular eosinophilic debris in the background. No necrotic debris or mucin was appreciated. Smears consisted of a mixed population of epithelial cells with abundant cytoplasm which appeared either granular and/or vacuolated. The cells with abundant granular cytoplasm had prominent nucleoli and binucleation without overt features of malignancy, resembling apocrine metaplasia, seen in breast FNA. The cells with abundant flocculent to vacuolated cytoplasm were somewhat reminiscent of sebaceous cells.
Pap-stained smears were also hypercellular and had a grungy background of debris. Smears also revealed the aforementioned two cell populations of either vacuolated or apocrine/oncocytic cells. Vacuolated epithelial cells were either microvesicular or contained abundant wispy cytoplasm. These cells had round to oval nuclei with finely granulated chromatin and smooth membranes and lacked prominent nucleoli. The apocrine cells had a round nucleus with a single prominent nucleoli and abundant granular cytoplasm. However the nuclear to cytoplasm ratio was low.
Histology
Sections from the subsequent resection specimen showed a circumscribed lobular mass composed of a mixture of increased glandular and ductal components with traversing bands of sclerosis. Acinar cells contained large, brightly eosinophilic cytoplasmic granules of various sizes, resembling zymogen granules of the pancreas. The ductal component had prominent apocrine and sebaceous metaplasia. Centrally located in the lesion is a dense fibrotic stroma causing cystic dilatation peripherally to the mass resembling sclerosing adenosis of the breast.There is limited literature on FNA cytologic diagnosis of SPA.(5-8) Some authors indicated that the presence of mixed population of apocrine-like and sebocyte-like cells is highly suggestive of SPA. Further, it was emphasized that the suspicion of SPA should be even stronger if the above cytopathological findings are seen in conjunction with intraductal epithelial proliferation; however, other findings have also been described in individual cases, including foamy histiocytes, lymphoplasmacytic components, neutrophils, squamoid cells, degenerated epithelial cells, and necrotic debris. Due to the diversity in cytomorphology, one should consider differential diagnoses which include but are not limited to sialadenitis, oncocytoma, Warthin’s tumor, mucoepidermoid carcinoma and acinic cells carcinoma. In this regard, abundant cells with acinar differentiation may result in difficulty in distinguishing SPA from acinic cell carcinoma. Also, the presence of a ‘‘dirty’’/cystic background may raise the possibility of a
In summary, the histologic features of SPA are highly-characteristic, arriving to a correct diagnosis on resection is challenging due to its rarity. Furthermore, cytopathologic features demonstrate a wide spectrum of cells suggestive but not diagnostic of SPA, making this diagnosis extremely challenging. Cytopathologists should be aware of this entity and avoid misinterpretation of SPA as a malignancy in the pre-operative workup of a parotid mass.
Warthin’s tumor or low grade mucoepidermoid carcinoma.Answers:
1. E
2. B
3. B
4. CReferences:
1. Smith BC, Ellis GL, Slater LJ, Foss RD. Sclerosing polycystic adenosis of major salivary glands. A clinicopathologic analysis of nine cases. Am J Surg Pathol. 1996;20(2):161-70.
2. Petersson F. Sclerosing polycystic adenosis of salivary glands: a review with some emphasis on intraductal epithelial proliferations. Head Neck Pathol. 2013;7 Suppl 1:S97-106.
3. Skalova A, Gnepp DR, Simpson RH, Lewis JE, Janssen D, Sima R, et al. Clonal nature of sclerosing polycystic adenosis of salivary glands demonstrated by using the polymorphism of the human androgen receptor (HUMARA) locus as a marker. Am J Surg Pathol. 2006;30(8):939-44.
4. Canas Marques R, Felix A. Invasive carcinoma arising from sclerosing polycystic adenosis of the salivary gland. Virchows Arch. 2014;464(5):621-5.
5. Etit D, Pilch BZ, Osgood R, Faquin WC. Fine-needle aspiration biopsy findings in sclerosing polycystic adenosis of the parotid gland. Diagn Cytopathol. 2007;35(7):444-7.
6. Fulciniti F, Losito NS, Ionna F, Longo F, Aversa C, Botti G, et al. Sclerosing polycystic adenosis of the parotid gland: report of one case diagnosed by fine-needle cytology with in situ malignant transformation. Diagn Cytopathol. 2010;38(5):368-73.
7. Gupta R, Jain R, Singh S, Gupta K, Kudesia M. Sclerosing polycystic adenosis of parotid gland: a cytological diagnostic dilemma. Cytopathology. 2009;20(2):130-2.
8. Imamura Y, Morishita T, Kawakami M, Tsuda G, Fukuda M. Sclerosing polycystic adenosis of the left parotid gland: report of a case with fine needle aspiration cytology. Acta cytologica. 2004;48(4):569-73.Contributed by:
Jennifer Hipp, M.D., Ph.D.
Fellow, Surgical Pathology
Department of Pathology
University of Michigan
Ann Arbor, Michigan, USAXin Jing, M.D.
Associate Professor
Director, Cytopathology Fellowship Program
Department of Pathology
Ann Arbor, Michigan, USA