Case of the Month ...
Clinical History:
24 year-old man with chief complaint of steatorrhea for four months. Cross-sectional imaging showed an ill-defined, hypovascular 1.0 cm mass in the pancreatic head with upstream pancreatic duct dilatation. An EUS-FNA was performed.
Diagnosis & Discussion
click on image for larger version
Figure 4 Figure 5 Images 1-5:
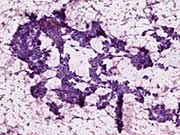
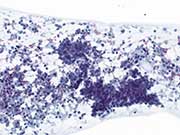
Figure 1: EUS-FNA, Alcohol fixed Papanicolaou stain
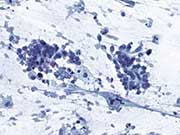
Figure 2: EUS-FNA, Alcohol fixed hematoxylin and eosin stain
Figure 3: EUS-FNA, Alcohol fixed Papanicolaou stain
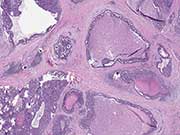
Figure 4. Resection, H&E stain, low magnification
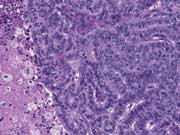
Figure 5. Resection, H&E stain, high magnificationFigure. 1. Small, epithelioid cells are present in hyperchromatic, three dimensional clusters within which tubular/acinar arrangements can be identified. There is a background of histiocytes, blood, and necrosis
Figure 2. The cells have eosinophilic cytoplasm with poorly defined borders.
Figure. 3. Nuclei are small and ovoid with inconspicuous nucleoli, fine chromatin, and minor nuclear membrane irregularities.
Figure 4. An intraductal proliferation with mostly tubular architecture and central comedo-like necrosis expands the ducts. The peri-ductal stromal response can make the determination of invasion very difficult.
Figure 5. Tubular arrangements of cuboidal to columnar cells with ovoid nuclei from the resected ITPN.
Questions:
What is the diagnosis?
- Adenocarcinoma
- Acinar cell carcinoma
- Intraductal papillary mucinous neoplasm, high grade
- Intraductal tubulopapillary neoplasm
- Poorly differentiated neuroendocrine carcinoma, large cell type
- The most likely immunophenotype for this neoplasm is:
- CK19 (+), CK7 (+), SMAD4 (-)
- MUC1 (+), MUC6 (+), MUC5AC (-), MUC2 (-)
- MUC2 (+), CDX2 (+), CK20 (+)
- Synaptophysin (+), Chromogranin (+), INSM1 (+), Rb1 (-)
- Trypsin (+), chymotrypsin (+), BCL10 (+)
- Select the true statement from the following:
- Acinar cell carcinoma and poorly differentiated neuroendocrine carcinoma are in the morphological differential diagnosis with intraductal papillary mucinous neoplasm
- Intraductal tubulopapillary neoplasm has similar recurrent mutations to conventional ductal adenocarcinoma.
- Invasion is rarely associated with intraductal tubulopapillary neoplasm.
- Papillary architecture is the most prominent cytoarchitectural feature of ITPN.
- The prognosis of intraductal tubulopapillary neoplasm associated with invasive carcinoma is similar to conventional ductal adenocarcinoma when invasion is detected histologically.
Discussion:
Intraductal tubulopapillary neoplasm (ITPN) is a rare entity arising within the pancreatic ducts that has a distinct morphological appearance compared with other intraductal neoplasms.1-3 The ducts are expanded by compact tubular arrangements of cells with occasional papillae. The mean age of clinical presentation is in the mid sixth decade with no sex predilection. The radiologic appearance is solid and focally cystic, but diffuse duct involvement can occur. In contrast to the dismal prognosis of conventional ductal carcinoma, patients with exclusively intraductal disease have 100% survival at five years. Invasion is seen in up to 70% of ITPN in the largest published series.2Patients with an associated invasive carcinoma have a 71% five-year survival.2
The cytomorphological appearance is distinctive for high cellularity with crowded, cohesive aggregates of cells arranged in closely packed tubules, solid clusters, and sheets.4-6 Despite the name, papillary features are not typically prominent on cytology or histology. On smears, the clusters have a hyperchromatic low power appearance. In some cases, the cells are monotonous, but pleomorphism is also possible. Ovoid nuclei are most common, with fine chromatin, mild membrane irregularities and inconspicuous nucleoli. On H&E preparations, an eosinophilic to amphophilic hue to the cytoplasm can be appreciated. ITPN with clear cells has been described. The background often contains necrosis, histiocytes, and blood with an absence of mucin.
The immunophenotype of ITPN is distinct from other intraductal neoplasms. Cytokeratins expressed include CK7, CK8, CK18, and CK19. The majority express MUC1 and MUC6 while MUC5AC and MUC2 are negative. Labeling for CDX2, synaptophysin, chromogranin, trypsin, and chymotrypsin is rare. 2
The entity with the closest cytological resemblance to ITPN is acinar cell carcinoma, which in rare instances has a primarily intraductal presentation. Pancreatoblastoma can occur in adults on rare occasion, and given the youth of this individual, it was entertained as a possibility. The presence of rosette-like tubules raised the differential diagnosis of a neuroendocrine neoplasm and the background necrosis suggested a high grade. Given this differential, it is helpful to know that ITPN is generally negative for immunohistochemical stains for acinar and neuroendocrine differentiation and beta-catenin (nuclear). The differential diagnosis also includes intraductal papillary mucinous neoplasm but the staining pattern is different from ITPN. Specifically, the pancreatobiliary type of IPMN labels with MUC1, MUC6 and MUC5AC and intestinal type IPMN labels with CDX2 and MUC2. One stain that could be used in the differential diagnosis with conventional ductal adenocarcinoma is SMAD4 because staining is lost in approximately 55% of adenocarcinomas, but is not expected to be lost in ITPN. The pronounced tubulo-glandular architecture and cytologic atypia of ITPN makes solid pseudopapillary neoplasm an unlikely entity in the differential diagnosis.
Molecular analysis has shown that ITPN lacks the common mutations of conventional ductal adenocarcinoma and IPMN including TP53, SMAD4, KRAS, GNAS, and RNF43. The genes with recurrent mutations include p16/CDKN2A, chromatin remodeling genes (MLL1, MLL2, MLL3, BAP1, PBRM1, and ATRX), members of the phosphatidylinositol 3-kinase pathway (PIK3CA, PIK3CB, and PTEN), and FGFR2 fusions.7
Given the rarity of this neoplasm, it is very challenging to make the specific diagnosis based on cytology alone and it is reasonable to provide diagnosis of an epithelial neoplasm with at least high grade dysplasia. Excluding entities such as acinar cell carcinoma and neuroendocrine carcinoma with immunohistochemistry can also be helpful.Answers:
1. D
2. B
3. AReferences:
- Yamaguchi H, Shimizu M, Ban S, et al. Intraductal tubulopapillary neoplasms of the pancreas distinct from pancreatic intraepithelial neoplasia and intraductal papillary mucinous neoplasms. Am J Surg Pathol. 2009;33:1164-1172.
- Basturk O, Adsay V, Askan G, et al. Intraductal tubulopapillary neoplasm of the pancreas: A clinicopathologic and immunohistochemical analysis of 33 cases. Am J Surg Pathol. 2017;41:313-325.
- Tajiri T, Tate G, Inagaki T, et al. Intraductal tubular neoplasms of the pancreas: Histogenesis and differentiation. Pancreas. 2005;30:115-121.
- Tajima S. Intraductal tubulopapillary neoplasm of the pancreas suspected by endoscopic ultrasonography-fine-needle aspiration cytology: Report of a case confirmed by surgical specimen histology. Diagn Cytopathol. 2015;43:1003-1006.
- Savant D, Lee L, Das K. Intraductal tubulopapillary neoplasm of the pancreas masquerading as pancreatic neuroendocrine carcinoma: Review of the literature with a case report. Acta Cytol. 2016;60:267-274.
- Furuhata A, Minamiguchi S, Mikami Y, et al. Intraductal tubulopapillary neoplasm with expansile invasive carcinoma of the pancreas diagnosed by endoscopic ultrasonography-guided fine needle aspiration: A case report. Diagn Cytopathol. 2014;42:314-320.
- Basturk O, Berger MF, Yamaguchi H, et al. Pancreatic intraductal tubulopapillary neoplasm is genetically distinct from intraductal papillary mucinous neoplasm and ductal adenocarcinoma. Mod Pathol. 2017;30:1760-1772.2001;7(9):351-3.
Contributed by:
Carlie Sigel, MD
Department of Pathology
Memorial Sloan-Kettering Cancer Center