Case of the Month ...

A 52-year-old female presented with a right acetabular mass increasing in size, currently measuring 7.8 x 4.1 cm and associated with osseous destruction. The patient has a history of an expansile lytic lesion previously diagnosed as a giant cell tumor of bone on biopsy 2 years ago. Fine needle aspiration of the new lesion is performed.
Authors
- Daisy Sun, MD PhD, Miami Valley Hospital, Dayton, OH
- Mia Wang, The Seven Hills School, Cincinnati, OH
click on image for larger version
Figure 3 Figure 4 Figure 5 Figure 6 Images 1-6:
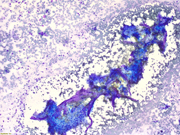
- Figure 1: Rapid on-site evaluation (ROSE) FNA material of the right acetabular mass, smear, Diff-Quik stain, 40x magnification.
- Figure 2: Rapid on-site evaluation (ROSE) FNA material of the right acetabular mass, smear, Pap stain, 40x magnification.
- Figure 3: Rapid on-site evaluation (ROSE) FNA material of the right acetabular mass, smear, Diff-Quik stain, 100x magnification.
- Figure 4: Rapid on-site evaluation (ROSE) FNA material of the right acetabular mass, smear, Pap stain, 100x magnification.
- Figure 5: Rapid on-site evaluation (ROSE) FNA material of the right acetabular mass, smear, Pap stain, 100x magnification.
- Figure 6: Core biopsy of the right acetabular mass lesion, H&E, 100x magnification.
Questions:
What is the diagnosis?
- Chondroblastoma
- Giant cell tumor of bone
- Giant cell tumor of bone after treatment with denosumab
- Osteosarcoma
- Which of the following molecular aberrations is most often seen in giant cell tumor of bone (GCTB)?
- H3F3A
- P53
- HRAS
- K36M
- Which of the following is true about giant cell tumor of bone (GCTB)?
- GCTB is composed of neoplastic osteoclast-like giant cells and nonneoplastic mononuclear stromal cells
- GCTB is positive for HG34W
- GCTB treated with denosumab shows a striking increase of osteoclast-like giant cells
- GCTB typically affects the diaphysis of long bones
Answers:
Question 1: Correct answer is C
The smears (Figures 1-5) show a cellular lesion composed of cytologically bland spindled cells, either in cohesive clusters or dispersed in the background. Some of the cohesive cellularity is associated with bone deposition as well as metachromatic stroma. The background mononuclear cells are oval to spindled. Strikingly, no single multinucleated giant cell is identified. The core biopsy shows a lesion with decreased cellularity, myxoid background and bland spindled cells. Again, no multinucleated giant cells are appreciated. The morphology is classic for giant cell tumor of bone (GCTB) after treatment with denosumab. The classic histologic features of denosumab-treated GCTB are reduction in multinucleated giant cells, proliferation of mononuclear cells, and replacement of tumor by fibrous and osseous tissue.Question 2: Correct answer is A
GCTB is a locally aggressive and rarely metastasizing neoplasm. It represents 4-10% of primary bone tumors and 15-20% of benign bone tumors. Although benign, GCTB can locally recur, rarely transform into a malignant tumor, or metastasize to the lung. Radiologically, GCTB usually presents as an expansile, radiolucent, and osteolytic lesion, primarily affecting the epiphysis of the long bone. Histologically, GCTB is highly cellular and composed of three types of cells including neoplastic mononuclear stromal cells (MNSC), mononuclear macrophages, and osteoclast-like multinucleated giant cells (MNGC). The MNSCs can be plump, oval, or spindled, while the MNGCs have numerous centrally-located, oval-shaped, and compact nuclei. MNGCs are distributed throughout the lesion with variable concentrations and are a distinct feature of GCTB.
More than 90% of GCTB harbor mutations in the H3F3A gene. These mutations are restricted to the MNSC component and are absent in the giant cell component. The differential diagnosis of GCTB includes all the giant cell rich lesions of bone such as chondroblastoma, aneurysmal bone cyst, non-ossifying fibroma, osteosarcoma, and others. H3G34W antibody is a valuable diagnostic tool to distinguish GCTB from other mimics; however, H3G34W does not distinguish benign from malignant GCTB.
Question 3: Correct answer is B
The receptor activator of the nuclear factor kappa β (RANK) pathway is implicated in the pathogenesis of GCTB and plays a crucial signaling pathway role in bone remodeling. Normally, cell-to-cell contact and interaction between RANK and RANK ligand (RANKL) promotes osteoclast formation. Studies have shown that MNSCs in GCTB express high levels of RANKL which stimulate the recruitment of osteoclastic MNGCs from normal monocytic pre-osteoclast cells. MNGCs overexpress RANK. The osteoclastic MNGCs then absorb host bone which leads to the osteolysis associated with GCTB.
The treatment of GCTB is a local intralesional surgical approach, including curettage, curettage and bone grafting, and resection. Systemic therapy and irradiation are not standard of care due to the benign nature of GCTB. Denosumab has emerged as an option for advanced GCTB.
Denosumab is a human monoclonal antibody that specifically targets RANKL. By binding RANKL on MNSC, it prevents its interaction with RANK on osteoclast-like MNGC and its precursors, thus, effectively inhibiting differentiation, activation, and survival of osteoclast-like MNGC and its associated osteolysis. It shifts the dynamics of bone formation away from RANK-mediated osteoclastic bone reabsorption toward bone formation. Denosumab inhibits the neoplastic MNSC without inducing apoptosis, leading to its uncontrolled proliferation. The MNSC, the true neoplastic cell, differentiates to osteoblastic phenotype, leading to deposition of newly formed osteoid/woven bone after treatment. Histologically, after denosumab treatment, osteoclast-like MNGCs are strikingly depleted, like our case. A partial maturation of neoplastic stromal cells towards an osteoblastic phenotype occurs, and fibrous and osteoid matrix production is seen.REFERENCES:
Roitman PD, Jauk F, Farfalli GL, Albergo JI, Aponte-Tinao LA. Denosumab-treated giant cell tumor of bone. Its histologic spectrum and potential diagnostic pitfalls. Hum Pathol, 2017 May 63:98-97.
- Abbasi AN, Qamar J, Habib A, Ebad Ali SM, Ahmed S, Khan MW, Unraveling the mystery: A comprehensive review of multidisciplinary strategies for managing giant cell tumor of the bone, J of Orthopedic Reports, 2025 March
- Borkowska AM, Szumera-Cieckiewicz A, Szostakowski B, Pienkowski A, Rutkowski RL, Denosumab in giant cell tumor of bone: multidisciplinary medical management based on pathophysiological mechanisms and real-world evidence, Cancers (Basel), 2022 May, PMID: 35565419
- Hosseinzadeh S, Tiwari V, De Jesus O, Giant cell tumor (Osteoclastoma), StatPearls,2025 Jan