Case of the Month ...

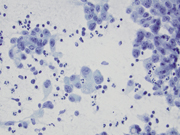
A 90-year-old female (former smoker with 5 pack years smoking history) with a 4.6 x 2.6 cm left lower lobe lung nodule underwent transbronchial fine-needle aspiration (FNA) for diagnostic evaluation. Cytology material was obtained for Diff-Quik–stained smears and cell block preparation. A concurrent core biopsy was performed for histologic correlation. Cytology smear and cell block material as well as representative immunohistochemical stains are below. Not pictured are TTF1 and p40 immunostains, which were both negative within tumor cells.
Authors
- Arkar Htoo, MD, FCAP. Cytopathology Fellow. Robert J. Tomsich Department of Pathology & Laboratory Medicine, Diagnostic Institute, Cleveland Clinic Foundation
- Rema Chaari, MD. Co-Section Head, Cytopathology, Robert J. Tomsich Department of Pathology & Laboratory Medicine, Diagnostic Institute, Cleveland Clinic Foundation
click on image for larger version
Figure 3 Figure 4 Figure 5 Figure 6 Images 1-6:
- Figure 1: Transbronchial FNA of left lower lobe lung nodule, aspirate smear, Diff-Quik stained, 20× magnification.
- Figure 2: Transbronchial FNA of left lower lobe lung nodule, aspirate smear, Pap stained, 20× magnification.
- Figure 3: Cell block, H&E stain, 20× magnification.
- Figure 4: Cell block, AE1/3 immunohistochemical stain, 10× magnification.
- Figure 5: Cell block, SMARC A4/BRG1 immunohistochemical stain, 20× magnification.
- Figure 6: Cell block, SMARC A2 immunohistochemical stain, 20× magnification
Questions:
- What is the most appropriate adequacy interpretation for this specimen?
- Non-diagnostic
- Atypical cells present
- Positive for malignancy
- Positive for non-small cell lung carcinoma
- Positive for adenocarcinoma with mucinous features
- What initial immunohistochemical panel would you perform?
- Synaptophysin, chromogranin, and INSM1
- TTF-1 and p40
- AE1/AE3 and Claudin-4 PAX8 and CK7
- Calretinin, D2-40, and HEG1
- Which immunohistochemical finding is most supportive of the final diagnosis?
- TTF-1 positivity
- p40 positivity
- Diffuse neuroendocrine marker expression
- Loss of SMARCA4 with retained SMARCA2
- Loss of SMARCB1 (INI1)
Answers:
Question 1: Correct answer is D. Positive for non–small cell lung carcinoma.
The specimen is adequately cellular and shows unequivocal malignant cytologic features, including pleomorphic high grade appearing malignant cells with nuclear enlargement with irregular contours, prominent nucleoli, and abnormal chromatin. Tumor cells also exhibited eccentrically placed round to irregular nuclei, finely textured nuclear chromatin, prominent nucleoli, and moderately abundant translucent to foamy cytoplasm in subsets of cellularity. These malignant cells are arranged in honeycomb-like sheets, three-dimensional clusters, acini, and occasional papillary structures. Cytoplasmic mucin vacuoles are present, with occasional cells showing a signet ring–like appearance.
At the time of adequacy assessment, it is best practice to distinguish small cell carcinoma from non–small cell carcinoma, as this distinction has immediate clinical and triage implications.
Although cytoplasmic mucin vacuoles are present and are suggestive of adenocarcinoma, mucinous vacuoles are not entirely specific and may also be encountered in other entities, including epithelioid mesothelioma and poorly differentiated carcinomas. Therefore, rendering a definitive diagnosis of adenocarcinoma at the time of adequacy without immunohistochemical confirmation is not ideal.
For these reasons, “positive for non–small cell lung carcinoma” represents the most accurate and clinically appropriate adequacy interpretation. This approach preserves diagnostic accuracy while allowing for appropriate ancillary studies to achieve definitive subclassification.
Question 2: Correct answer is B. TTF-1 and p40.
In the evaluation of a malignant lung cytology specimen, the initial immunohistochemical (IHC) panel should focus on confirming non–small cell lung carcinoma (NSCLC) and establishing lineage, specifically distinguishing adenocarcinoma from squamous cell carcinoma. TTF-1 is the most sensitive marker for pulmonary adenocarcinoma, while p40 is the most specific marker for squamous cell carcinoma. Together, these stains form the recommended first-line IHC panel in small biopsies and cytology specimens.
This approach is consistent with established diagnostic algorithms, in which TTF-1–positive/p40–negative tumors are classified as NSCLC favor adenocarcinoma, and TTF-1–negative/p40–positive tumors favor squamous cell carcinoma. Importantly, cases that are negative for both TTF-1 and p40 fall into an NSCLC-NOS category and warrant additional downstream workup, including epithelial markers and, when appropriate, SMARCA4 (BRG1) immunohistochemistry.
Option A (synaptophysin, chromogranin, INSM1) is not appropriate as an initial panel because the cytomorphology does not suggest small cell carcinoma or another neuroendocrine neoplasm (i.e., no nuclear molding, crush artifact, or classic neuroendocrine chromatin).
Option C (AE1/AE3 and Claudin-4) confirms epithelial differentiation but does not provide lineage-specific information and is more appropriately used after TTF-1 and p40 are negative, particularly in poorly differentiated tumors.
Option D (PAX8 and CK7) is not indicated as an initial panel in a primary lung lesion and is typically reserved for evaluation of possible metastatic disease.
Option E (calretinin, D2-40, HEG1) represents mesothelial markers and is not appropriate as a first-line panel, as the morphology does not primarily suggest a mesothelial neoplasm.
Thus, TTF-1 and p40 represent the most appropriate and efficient initial immunohistochemical panel, guiding subsequent reflex testing and avoiding premature or non-informative studies.
Question 3: Correct answer is D. Loss of SMARCA4 with retained SMARCA2.
Loss of SMARCA4 (BRG1) expression (Figure 5) with retained SMARCA2 (BRM) (Figure 6) is a defining immunophenotypic feature of SMARCA4-deficient non–small cell lung carcinoma[R(1.1]. These tumors typically show poorly differentiated morphology, lack expression of conventional lineage markers such as TTF-1 and p40, and retain epithelial differentiation as evidenced by cytokeratin and/or Claudin-4 positivity (Figure 4). Histologic correlation from the concurrent core biopsy revealed adenocarcinoma, with mucicarmine highlighting intracytoplasmic mucin. The biopsy also demonstrated loss of SMARCA4 expression, confirming the cytologic impression.
The finding of SMARCA4 loss in the appropriate morphologic and immunophenotypic context is critical, particularly in TTF-1–negative and p40-negative NSCLC, as it allows distinction from other poorly differentiated carcinomas and from primary thoracic sarcomas. Retention of SMARCA2 further supports classification within the SMARCA4-deficient carcinoma spectrum rather than alternative SWI/SNF–deficient neoplasms.
SMARCA4-deficient NSCLC, represent an aggressive subset of NSCLC (~5% of NSCLC) that respond poorly to targeted treatment and usually present at advanced stages. They lack expression of conventional lineage markers such as TTF-1, p40 and p63 with retention of expression to keratins and claudin-4. Cytomorphologic clues that could raise this possibility include pleomorphic malignant cells with eccentrically placed nuclei (“rhabdoid”) and high-grade epithelioid appearance with easily identifiable mitoses and necrosis. Recognition of this entity on small biopsies and cytological specimens is critical to avoid misclassification and to ensure appropriate clinical management.
Option A (TTF-1 positivity) would favor conventional pulmonary adenocarcinoma and argues against a SMARCA4-deficient tumor, which is characteristically TTF-1–negative.
Option B (p40 positivity) supports squamous cell carcinoma and is incompatible with the final diagnosis.
Option C (diffuse neuroendocrine marker expression) would suggest small cell carcinoma or another neuroendocrine neoplasm; however, SMARCA4-deficient non small cell carcinomas typically lack diffuse neuroendocrine marker expression[R(2.1][AH2.2].
Option E (loss of SMARCB1 [INI1]) is characteristic of a different group of SWI/SNF–deficient tumors, including epithelioid sarcoma and malignant rhabdoid tumors, and is not a feature of SMARCA4-deficient non small cell lung carcinomas.
Overall, loss of SMARCA4 with retained SMARCA2, in conjunction with epithelial marker expression and negativity for TTF-1 and p40, is most supportive of the final diagnosis of SMARCA4-deficient non–small cell lung carcinoma.
References
- WHO Classification of Tumours Editorial Board. Thoracic Tumours. 5th ed. International Agency for Research on Cancer; 2021. WHO Classification of Tumours; vol 5. https://publications.iarc.fr/595
- Cibas ES, Ducatman BS, VanderLaan PA, eds. Cibas and Ducatman’s Cytology: Diagnostic Principles and Clinical Correlates. 6th ed. Elsevier; 2026.
- Mehta A, Bansal D, Tripathi R, et al. SMARCA4/BRG1 protein–deficient thoracic tumors dictate re-examination of small biopsy reporting in non–small cell lung cancer. J Pathol Transl Med. 2021;55(5):307–316.